Despite solid technology and competitive pricing, many Asian CDMOs struggle to win global clients. Here explores 5 deep-rooted structural barriers — regulatory hurdles, reputation building, geopolitical risks, communication challenges — prevent Asian CDMOs from achieving true globalization.

“Our technology is solid, our price is competitive—but why aren’t Western clients buying in?”
This is the question I hear most often from Asian CDMO business development teams at international conferences. Despite favorable conditions and competitive pricing, we’ve still often hit a wall. I bet you know this frustration if you worked in BD or global business development.
Through years of participating in international exhibitions and conducting partnership negotiations with companies worldwide, I’ve discovered that Asian regional CDMOs (Contract Development and Manufacturing Organizations) face not merely a “marketing problem”, but several deep-rooted structural barriers.
This article objectively examines the five major challenges that Asian CDMOs, particularly small to medium-sized players, encounter in their globalization journey from a practical perspective.
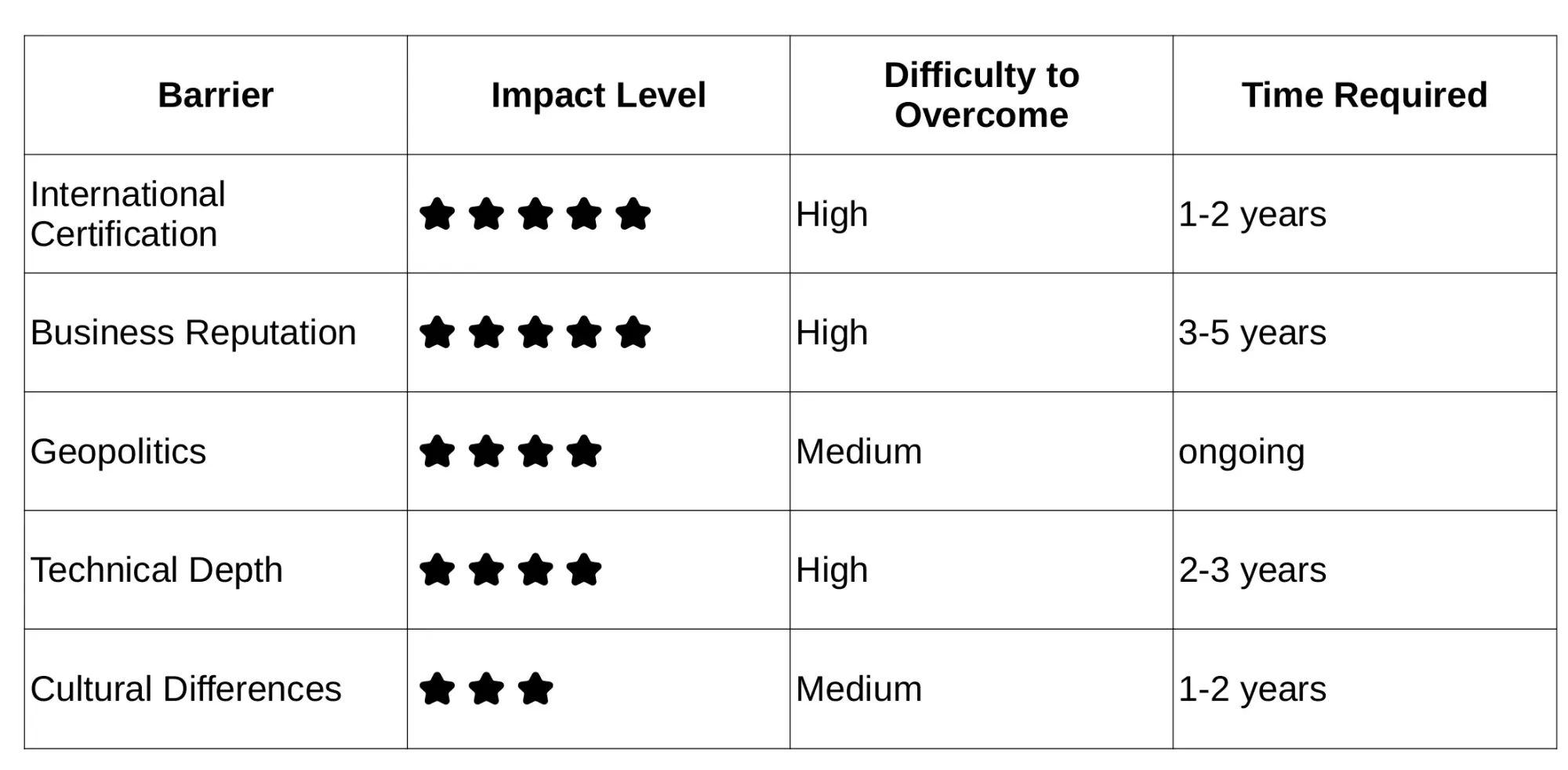
The pharmaceutical industry ranks among the world’s most heavily regulated sectors. Drug safety issues directly threaten public health and undermine national productivity and competitiveness, making regulatory oversight extremely strict and rigorous.
To enter major markets like the US and Europe, pharmaceuticals must have two certificates:
This is what the pharmaceutical industry refers to as requiring both “drug license” and “facility certificate”—both are indispensable.
You might think: “If I only target clinical-stage drug collaborations, I can avoid this issue, right?”
Theoretically, yes—clinical-stage investigational drugs only require GMP-compliant manufacturing processes. But here’s the reality: when your competitors already hold FDA, EMA, or Japan PMDA approved GMP certificates, your attractiveness plummets.
Why?
Establishing and maintaining quality management systems meeting international standards requires:
For smaller CDMOs, this represents a formidable barrier.
When selecting CDMOs, Western clients prioritize not only quality, cost, and technical capabilities, but also business reputation and historical collaboration experience.
Clients are extremely concerned about confidentiality. While drugs themselves have patent protection, the technical know-how represents a key competitive barrier.
Asian small-to-medium CDMOs relatively lack long-term collaboration history with major international pharmaceutical companies, and trust takes time to build. This creates a vicious cycle:
No International Clients → No Collaboration Experience → Difficult to Earning Trust → No International Clients (again)
I understand this dilemma—all beginnings are difficult. But lacking collaboration experience doesn’t mean failure; rather, it means CDMOs need to work hard to overcome this obstacle and find their first client—opening the business from 0 to 1, then gradually progressing from 1 to 100.
Further Reading: See “Strategy 5: Build an Industry Network Systematically” in the article
Following COVID-19 and the US-China trade war, global enterprises increasingly emphasize supply chain stability and diversification to mitigate geopolitical risks or pandemic supply disruptions.
For international pharmaceutical companies, over-concentration in a single Asian country may be viewed as risky. Especially after US protectionism and trade wars, these factors influence client choices:
If a CDMO operates multiple manufacturing facilities across different countries, it can transfer production to other sites when problems arise—naturally more attractive to clients.
Practical Note: Facility transfers aren’t simply relocating; they require overcoming technical transfer and regulatory approval challenges, but at least provide backup options.
International situations continuously evolve, and clients’ outsourcing strategies adjust accordingly. CDMOs need flexibility to respond to geopolitical changes.
Whether small molecule drugs, protein therapeutics, mRNA vaccines, or continuously innovating emerging therapies (gene therapy, cell therapy), these technologies primarily originate from Western R&D innovation.
Leading Western CDMOs, having participated earlier, typically possess:
For Asian small-to-medium CDMOs, sustained investment is needed to narrow the technical and experience gap.
Some might say: “From this perspective, Asian small-to-medium CDMOs will forever be playing catch-up, never winning!”
I can only say this is an accumulation process. CDMOs need to accumulate manufacturing technology experience and capabilities. Once sufficient know-how is accumulated:
Further Reading: See “Strategy 1: Focus and Deepen in a Specific Niche Market” in the article
Working with foreign companies presents certain challenges that require international talent to manage. However, working across different time zones presents challenges:
These factors create collaboration challenges and factors, and clients need to evaluate them in partnership.
- Problem description
- Possible solutions
- Risk assessment
- Cost analysis
- Discussion
Not every European country’s native language is English; that means both sides may struggle in communication and discussion.
If each party’s English communication conveys only 85% of the actual meaning, final understanding drops to just 72% (0.85×0.85).
This not only delays problem resolution but may cause misunderstandings leading to wrong decisions, affecting entire project timelines.
Commercial customs and interpersonal cultures vary across countries:
After reviewing these 5 barriers, you might think: “Is Asian CDMO globalization too difficult?” Indeed, these are structural barriers not overcome in the short term. But this doesn’t mean there’s no opportunity.
These companies rose by riding the wave of protein therapeutics trend, establishing strong footholds in international markets and proving Asian CDMOs can absolutely compete on the global stage.
The key is choosing the right strategies, finding the right entry points, and building competitive advantages.
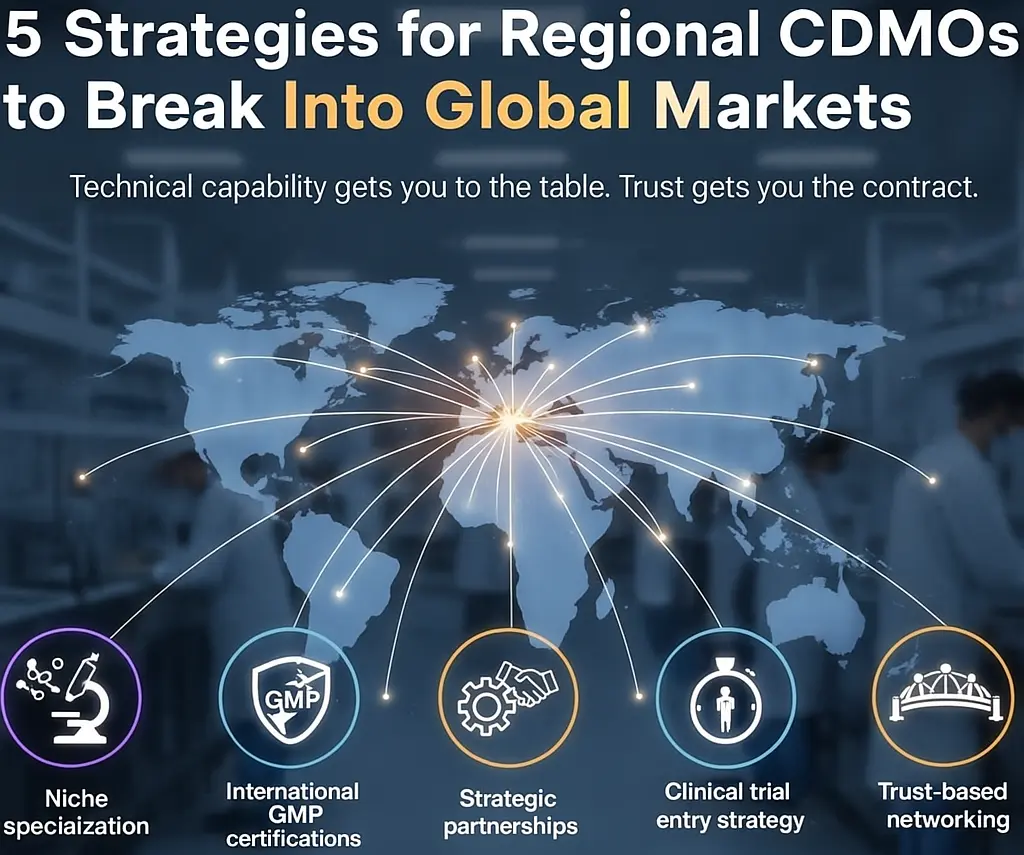
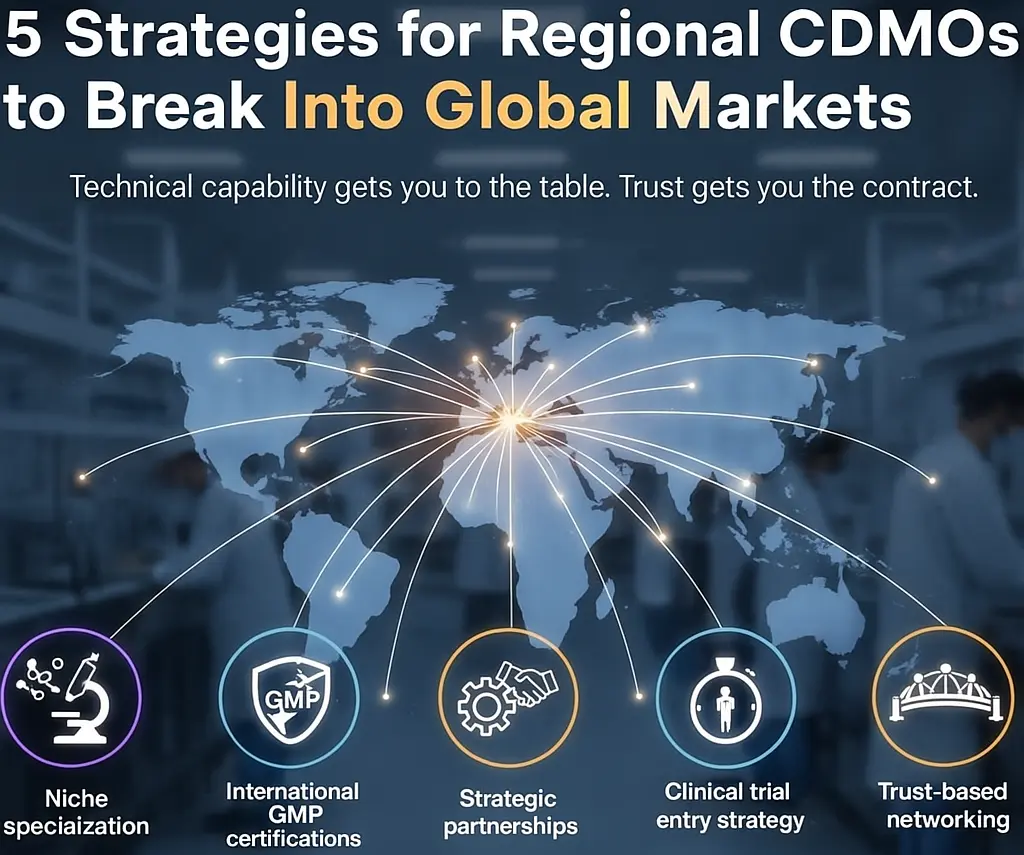
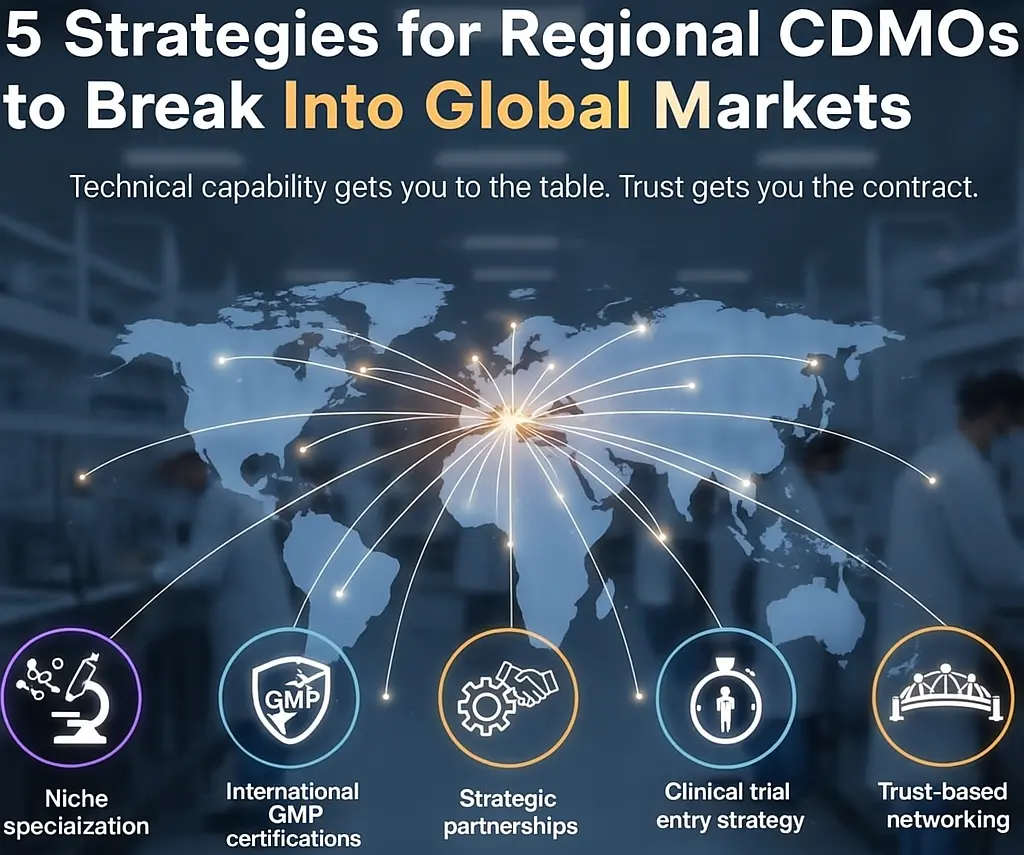
In the next article, “How Asian CDMOs Break into Global Markets: 5 Practical Strategies” I’ll share:
These strategies and practices of successful enterprises can serve as reference for small-to-medium CDMOs.
Please feel free to leave comments and connect me through Email and LinkedIn to share your experiences or challenges you’ve encountered in the business globalization!
Feel free to sign up for our newsletter!